
We are all fascinated by diamonds. I have given my beloved wife some that sparkle with a range of colours in a totally unique way. But had we lived around 1900, these diamonds would not have been so bright.
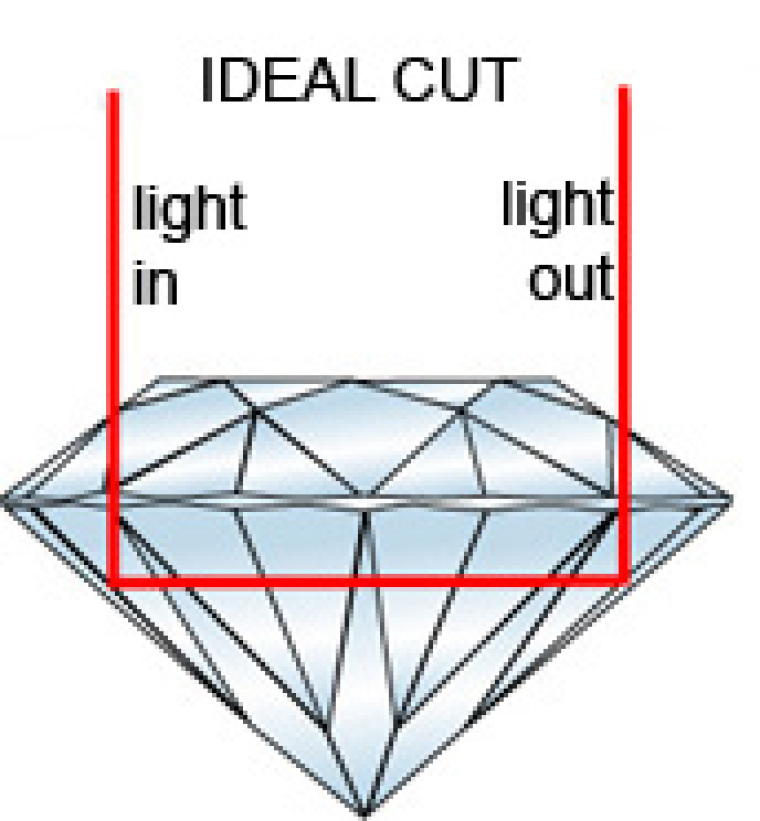
One of my relatives inherited a beautiful and quite large stone from her grandmother, but it looks slightly milky and not so shiny as my wife's more modern (but smaller) ring. A jeweller explained that, before they understood the physics of the way the light passes through the crystalline stone, they cut the rough diamonds with 1/3 above the plane (the girdle).
But that is not the optimum angle, it is too shallow. They now know that the maximum sparkle from the re-reflection of light is achieved with about 15% of the stone above the girdle. (See accompanying photo from http://www.makermends.com/diamonds.html)



A science lesson about diamonds (be warned: technical language follows)
If you look carefully at a cut diamond, you will see that it has many little flat faces (facets), and each one can refract and reflect light. The reason that a rough diamond can be easily split into a lovely jewel is because the carbon atoms in the diamond stone are arranged in a regular pattern of tiny, tiny tetrahedral (four-sided) shapes. When millions of these atoms build one upon the other to make the larger stone, the structure is retained in a larger form and we see the tetrahedral faces that with our eyes.
This sub-microscopic structure was first shown by Australian father and son Nobel laureates in 1915, Lawrence Bragg and William Bragg, using their then new technique of X-ray crystallography. Instead of taking an X-ray picture of bones, they fine-tuned their machine to bounce the X-rays off microscopic structures, and then analysed the reflected pattern mathematically to reconstruct the original shape of the tiny crystals and molecules. The attached photo shows what they found for the carbon atoms in diamond.

Diamonds are formed a hugely long way down under the Earth where there are high temperatures and pressures. This causes the carbon atoms to be squeezed tightly together, and also vaporises any other fossilised tissue and minerals. Where there has once been an eruption of hot materials from deep under the surface, "pipes" that contain these diamonds are brought to the surface. There are only a few places in the world where these have emerged; however now there is also a newer process of retrieving them from the sea bed in a few selected areas.
It is said: "a diamond is just a piece of charcoal that handled stress extremely well."
The chemical nature of carbon atoms is such that they can form four very strong bonds with other atoms; in a diamond of course, these bonds are all with other carbon atoms – nothing else (represented by the black balls in the model). As well as enabling the cutting into sparkly jewels, these strong and stable tetrahedral structures make diamonds extremely strong and hard. Until a few artificial minerals were made (which mimic the structure of diamonds), diamond was the hardest substance known.

Not just a pretty face – hard-working diamonds
The only references to diamond in the Bible are to its use as a stylus to grind other materials. And the vast bulk of mined diamonds are so tiny that they look like dust. These are relegated to industrial uses such as grinding, engraving, abrasion and lubrication. I was amused to see in an advertisement that diamond-encrusted rollers are even used in a pedicure device to help keep people's feet smooth and attractive.
Only a very, very small proportion of diamonds are actually large enough and of good enough quality (without flaws) to be cut and polished into jewellery – this is why they are so very rare, and therefore so very expensive.
And because these 4 bonds are so strong, diamonds are almost totally inert and do not react with living tissue. This enables tiny little nano-diamonds to be used in medical treatment.
Making artificial diamonds
Current methods can make synthetic diamonds in a few days. The scientists either reproduce high pressures and temperatures in equipment in the lab, or else they use lasers to vaporise carbon-containing material and then "condense" the pure diamonds on a surface. Some of these can be made into jewellery but most are used in industrial processes similar to the fine diamond dust acquired from mining.
However, I was more interested in a few innovative ways of using tiny diamonds in medicine. This posed a reflective moment as I considered Romans chapter 12 verse 2: "Do not be conformed to this world, but be transformed by the renewal of your mind, that by testing you may discern what is the will of God, what is good and acceptable and perfect."

Uses in medical therapy and diagnosis
Tiny nano-diamonds can be slightly electrostatic, so cancer-fighting drugs can be encouraged to stick to their surface. Because the diamonds themselves are totally unreactive and not dangerous to the human body, researchers are looking at ways to use them to carry the drugs to the actual sites of cancer in human patients.
In Melbourne, researchers are looking at ways to use synthetic nano-diamonds to actually stimulate nerves "Professor Prawer, inaugural head of the Melbourne Materials Institute at Melbourne University, said 'We have discovered a form of diamond that ... we can put an electrical signal onto it that then causes the neurons to fire and get a response.'"
At Bionic Vision Australia, these will be used to encase a tiny chip that will be inserted into transplanted eyes in a forthcoming trial. If successful, they could be used to stimulate damaged nerves in patients with ailments such as Parkinson's disease.
Our dental health is also important, and dental researchers in America have discovered a way to make a much more bio-compatible paste or glue, to seal off nerve endings when people have root canal therapy.
Finally, back in Sydney, Australia, another group of medical researchers has teamed up with theoretical physicists, and found a way to make nano-diamonds into tiny magnets, and then use them to enhance MRI (magnetic resonance imaging) when detecting tumours and other diseases.
So it seems whichever way you cut it, diamonds may in the future become everyone's best friends.

Dr Mark Tronson is a Baptist minister (retired) who served as the Australian cricket team chaplain for 17 years (2000 ret) and established Life After Cricket in 2001. He was recognised by the Olympic Ministry Medal in 2009 presented by Carl Lewis Olympian of the Century. He mentors young writers and has written 24 books, and enjoys writing. He is married to Delma, with four adult children and grand-children.
Mark Tronson's archive of articles can be viewed at http://www.pressserviceinternational.org/mark-tronson.html